Pb No3 2 Oxidation Number
OXIDATION NUMBERS Computer
To calculate oxidation numbers of elements in the chemical chemical compound, enter information technology'southward formula and click 'Calculate' (for example: Ca2+, HF2^-, Fe4[Fe(CN)6]3, NH4NO3, so42-, ch3cooh, cuso4*5h2o).
| Element: | Cu | Southward | O | * | H | O |
| Oxidation number: | +2 | +6 | -2 | +one | -2 |
The oxidation state of an atom is the charge of this atom later on ionic approximation of its heteronuclear bonds. The oxidation number is synonymous with the oxidation land. Determining oxidation numbers from the Lewis structure (Figure 1a) is even easier than deducing it from the molecular formula (Effigy 1b). The oxidation number of each atom can be calculated by subtracting the sum of lone pairs and electrons it gains from bonds from the number of valence electrons. Bonds between atoms of the same element (homonuclear bonds) are ever divided equally.
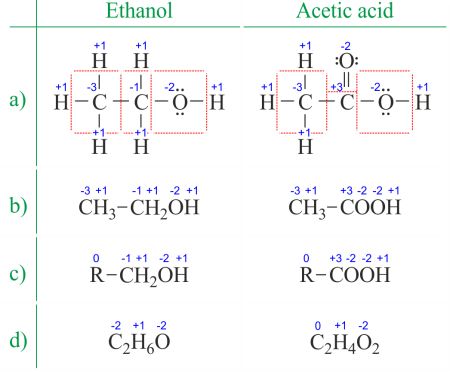
Figure 1. Different means of displaying oxidation numbers of ethanol and acetic acrid. R is an abridgement for any group in which a carbon atom is attached to the rest of the molecule past a C-C bond. Notice that changing the CH3 group with R does non change the oxidation number of the central atom. → Download high quality image
When dealing with organic compounds and formulas with multiple atoms of the aforementioned element, information technology's easier to piece of work with molecular formulas and average oxidation numbers (Figure 1d). Organic compounds tin be written in such a way that annihilation that doesn't alter before the commencement C-C bail is replaced with the abridgement R (Figure 1c). Unlike radicals in organic molecules, R cannot be hydrogen. Since the electrons betwixt 2 carbon atoms are evenly spread, the R group does not change the oxidation number of the carbon atom information technology's attached to. You lot can detect examples of usage on the Dissever the redox reaction into two half-reactions page.
Rules for assigning oxidation numbers
- The oxidation number of a free chemical element is always 0.
- The oxidation number of a monatomic ion equals the charge of the ion.
- Fluorine in compounds is always assigned an oxidation number of -ane.
- The alkali metals (group I) always have an oxidation number of +1.
- The alkaline earth metals (group Two) are always assigned an oxidation number of +2.
- Oxygen about e'er has an oxidation number of -ii, except in peroxides (H2O2) where it is -ane and in compounds with fluorine (OF2) where it is +two.
- Hydrogen has an oxidation number of +1 when combined with non-metals, but information technology has an oxidation number of -1 when combined with metals.
- The algebraic sum of the oxidation numbers of elements in a compound is zero.
- The algebraic sum of the oxidation states in an ion is equal to the charge on the ion.
Assigning oxidation numbers to organic compounds
- The oxidation state of whatsoever chemically bonded carbon may be assigned past adding -1 for each bond to more electropositive atom (H, Na, Ca, B) and +one for each bond to more electronegative atom (O, Cl, N, P), and 0 for each carbon atom bonded directly to the carbon of interest. For instance:
- propene: CH3-CH=CH2
- lauric acid: CH3(CH2)10COOH
- di-tert-butyl peroxide: (CH3)3COOC(CH3)3
- diisopropyl ether: (CH3)2CH-O-CH(CH3)two
- dibenzyl sulfide: (C6H5CH2)2S
- cysteine: HO2CCH(NH2)CH2SH
Citing this folio:
Generalic, Eni. "Oxidation numbers calculator." EniG. Periodic Table of the Elements. KTF-Split up, 27 Oct. 2022. Web. {Date of admission}. <https://world wide web.periodni.com/oxidation_numbers_calculator.php>.
Pb No3 2 Oxidation Number,
Source: https://www.periodni.com/oxidation_numbers_calculator.php?eq=cuso4%2A5h2o
Posted by: blueseentiourcio.blogspot.com


0 Response to "Pb No3 2 Oxidation Number"
Post a Comment